Name: Niacin
Class: Vitamin Supplement (NAD+ precursor supplement)
Alias(es): CAS# 59-67-6, Synonyms: Nicotinic Acid, Vitamin B3, Brand Names: Niacor, Niaspan
Background: Niacin, also known as vitamin B3, is a century-old nicotinamide adenine dinucleotide (NAD+) precursor supplement. NAD+ is a crucial molecule in cellular metabolism and respiration, and levels of NAD+ fall naturally with age[1]. Raising NAD+ (NAD+ repletion) is also known to activate sirtuins, which stabilize telomeres[2] and promote healthy aging by signaling nutrient scarcity and catabolism[3]. The efficacy of Niacin in raising NAD+ levels was demonstrated in a recent study, wherein Niacin raised NAD+ levels in healthy humans and in those with mitochondrial myopathy and conferred several health benefits (improved strength and muscle mass, and reduced adiposity). The takeaway is: niacin demonstrably raises NAD+ levels in humans[4], and has beneficial effects that are similar to other NAD+ supplements such as NR and NMN[4,5], in addition to being established as an effective LDL (“bad”) cholesterol-lowering medication[6], although it may cause more side-effects than other NAD+ supplements (see safety-concerns section).
Is there evidence it works in humans for aging?
NCT03973203: (Completed December 31, 2018) Title: The Effect of Niacin Supplementation on Systemic Nicotinamide Adenine Dinucleotide (NAD+) Metabolism, Physiology and Muscle Performance in Healthy Controls and Mitochondrial Myopathy Patients.
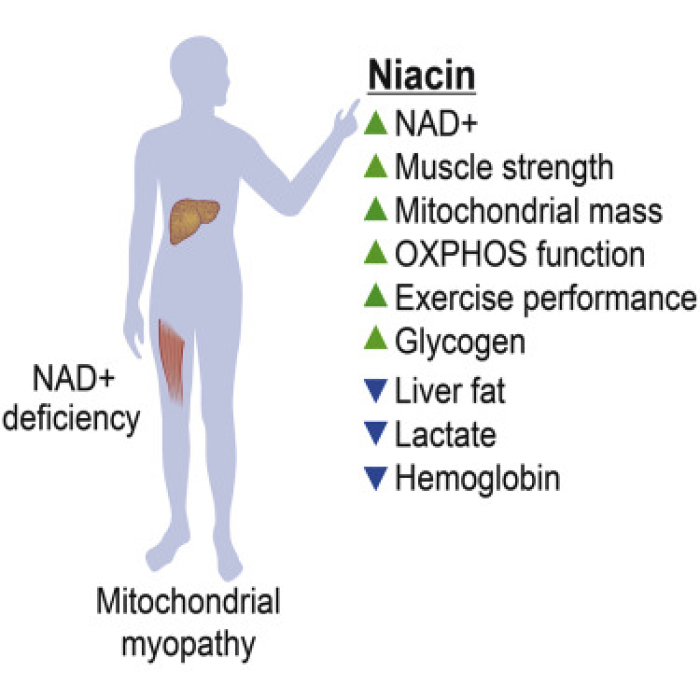
Results: Daily Niacin raises NAD levels in both healthy people and in those with mitochondrial myopathy, and confers several health benefits (improved strength and muscle mass, and reduced adiposity). This study involved niacin for a period of 10 months. These results can be found in an article by Pirinen et al., published in June 2020 in the journal Cell Metabolism. See: https://www.sciencedirect.com/science/article/pii/S155041312030190X
See figure from above paper:
Additionally, see table 1 below from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6342515/ for a list of human clinical trial outcomes of niacin for various age-related and non age-related diseases:
Is there evidence it works in preclinical studies for aging?
NA significantly extended lifespan of C. elegans, but suggested that may only be successful in individuals with low NAD levels (Yang et al., 2019).
See below Table 1 from a 2020 review article published in the journal Nature below for studies on nicotnamide, derivative of niacin. (The first reference is Mitchell et al., 2018, and the second is Liu et al, 2012.):
It is worth noting that mice do not require dietary niacin because they produce NAD+ through other biochemical pathways naturally. For this reason, studies involving niacin administration using mice as model organisms are not ideal. However, in 2018, a study by Palzer et al., published in the journal Cell Press describes a novel line of transgenic mice known as ANDY (acquired niacin dependency) mice that require dietary niacin and can utilize niacin in the same way humans do. It will be interesting to see new research on the effects of niacin administration in these transgenic mice, which will better model the potential benefits of niacin administration in humans as a treatment for declining NAD+ levels.
Are there known safety concerns?
The common Niacin side-effects, such as flushing, bloating, and tingling, are harmless and tend to go away with continued use[4,7,8]. Less common, but more serious side-effects of niacin, such as liver toxicity or damage, can occur at very high dosages of the sustained release tablets, but are not as common with the regular or extended-release supplements, especially at lower dosages[9]. Additionally, risk of diabetes, excess infections, and bleeding also increase moderately in those taking niacin with existing cardiovascular disease[10].
For more info on potential side-effects see: https://www.mayoclinic.org/drugs-supplements-niacin/art-20364984
Literature Cited:
1. Schultz, M. B., & Sinclair, D. A. (2016). Why NAD+ Declines during Aging: It’s Destroyed. Cell Metabolism, 23(6), 965–966. https://doi.org/10.1016/j.cmet.2016.05.022
2. Amano, H., Chaudhury, A., Rodriguez-Aguayo, C., Lu, L., Akhanov, V., Catic, A., Popov, Y. V., Verdin, E., Johnson, H., Stossi, F., Sinclair, D. A., Nakamaru-Ogiso, E., Lopez-Berestein, G., Chang, J. T., Neilson, J. R., Meeker, A., Finegold, M., Baur, J. A., & Sahin, E. (2019). Telomere Dysfunction Induces Sirtuin Repression that Drives Telomere-Dependent Disease. Cell Metabolism, 29(6), 1274-1290.e9. https://doi.org/10.1016/j.cmet.2019.03.001
3. López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2013). The Hallmarks of Aging. Cell, 153(6), 1194–1217. https://doi.org/10.1016/j.cell.2013.05.039